Researchers trial targeted proton beams to improve mesothelioma survival
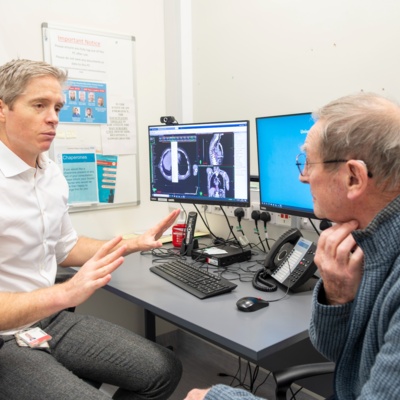
WHO: University College London Hospitals NHS Foundation Trust (UCLH)
WHAT: HIT-MESO randomised clinical trial investigating proton beam therapy (PBT)
WHERE: UCLH in London and The Christie NHS Foundation Trust in Manchester
WHEN: Trial details published 11 March 2026
WHY: To improve two-year survival rates for mesothelioma from 30% to 50%
The first UK mesothelioma proton beam therapy trial aims to transform outcomes for patients by delivering more precise radiation doses.
Launching the mesothelioma proton beam therapy trial
Clinician researchers at UCLH and UCL are leading a major UK study to treat mesothelioma. This rare and aggressive cancer affects the lining of the lung. It is usually caused by exposure to asbestos.
More than 2,700 people are diagnosed with the condition in the UK every year. Most of these patients are older men. Current treatment options are limited and there is no known cure.
Standard radiotherapy is often unsuitable for treating the full lung lining. Conventional beams risk damaging vital organs like the heart and liver. This can lead to serious side effects for the patient.
The HIT-MESO trial uses proton beam therapy instead. This technology uses targeted beams that deposit energy directly in the tumour. It minimises radiation exposure to surrounding healthy tissue.
Dr Crispin Hiley is the chief investigator of the HIT-MESO trial. He serves as a UCLH consultant oncologist. He explains that conventional radiotherapy cannot safely cover the full affected area.
Dr Crispin Hiley said: "Proton beam therapy allows us to deliver high-dose radiation far more precisely, sparing critical organs like the heart and healthy lung."
The trial aims to recruit up to 148 patients from 23 NHS centres. Participants receive daily treatment for five weeks. Fifty patients have already joined the study.
University College London Hospitals NHS Foundation Trust is one of the largest NHS trusts in the UK. It provides 1st class acute and specialist services to patients locally and nationally.
Peter Littlefield, 83, is one of the participants. He was diagnosed last autumn after experiencing a persistent cough. He travelled from Portsmouth to London for the five-week treatment block.
Peter added: "When they mentioned this trial using proton beam therapy, I said yes immediately. Being accepted was the best Christmas present I could have hoped for."
Peter is an active outdoorsman who enjoys fishing and shooting. He remains determined to stay mobile and positive throughout the process. He hopes the trial will give him more time to stay active.
The study is supported by the National Institute for Health and Care Research. Asthma + Lung UK is providing funding for the research. They describe the work as a real breakthrough.
Dr Samantha Walker, Director of Research and Innovation at Asthma + Lung UK, said: "If this trial is successful, it would offer new hope for the more than 2,000 people who are diagnosed with mesothelioma every year."
Researchers believe this approach could reshape national treatment guidelines. If survival rates increase as expected, it could fundamentally change mesothelioma care in the UK.
OFFICIAL SOURCE VERIFICATION:
This report is based on official clinical data from NHS Trust / Health Authority.
Document: **UCLH and UCL researchers trial proton beam therapy in bid to transform treatment for mesothelioma
Source Link: https://www.uclh.nhs.uk/news/uclh-ucl-researchers-trial-proton-beam-therapy-bid-transform-treatment-mesothelioma
Related Links
MORE: Over 10,000 Calls to Martha’s Rule Led to 1,885 Treatment Changes Across NHS Trusts — https://trendwiremedia.com/2026/03/09/over-10000-calls-to-marthas-rule-led-to-1885-treatment-changes-across-nhs-trusts/
MORE: NHS launches world-first DNA study to revolutionise mental health treatment — https://trendwiremedia.com/2026/02/14/nhs-launches-world-first-dna-study-to-revolutionise-mental-health-treatment/
MORE: Google AI Outperforms Doctors in Largest NHS Breast Cancer Screening Study Across UK — https://trendwiremedia.com/2026/03/10/google-ai-outperforms-doctors-in-largest-nhs-breast-cancer-screening-study-across-uk/
Subscribe for $2 every four weeks for the first six months
Subscribe for $20 every four weeks for the first six months